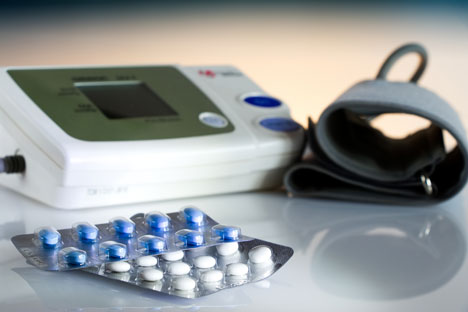
New Delhi: A three-in-one pill containing low doses of three medicines may be a more effective way to treat high blood pressure than conventional medication, health researchers said on Tuesday.
A clinical trial - conducted in Sri Lanka after drug regulators in India repeatedly rejected it - has found that the so-called triple pill helped 70 per cent of patients reach blood pressure targets compared with about half of those who received the standard medication.
The findings were published on Tuesday in the Journal of the American Medical Association.
In standard therapy, patients typically start treatment with a low dose of one drug, but the dose is increased or additional drugs are prescribed to achieve the blood pressure target. Patients are recalled to clinics at frequent intervals to see if the medication is working or to tailor the doses.
"We know many doctors and patients find it too complicated and often don't stick to the process - the new approach is simpler and it works," Ruth Webster, chief scientist at the George Institute for Global Health in Australia who led the study, said.
The triple pill contains three drugs - 2.5mg amplodipine, 12.5mg chlothalidone and 20mg telmisartan - each at half the standard dose for high blood pressure. The trial found that a significantly higher proportion of patients who received the triple pill responded adequatelycompared with those who received standard therapy.
After six months of therapy, over 80 per cent of patients on the triple pill had successful blood pressure control compared with the majority of patients who received standard therapy -a single drug or two or more blood pressure lowering drugs.
"We believe the results of the trial should influence clinical guidelines towards recommending initial or early use of combination therapy to treat even mild or moderate hypertension," Anushka Patel, chief scientist at the George Institute and study team member, told The Telegraph.
Current guidelines suggest patients with hypertension be started on one drug at a low dose which should be slowly increased if blood pressure targets are not met. Then, additional drugs are introduced if targets are still not met.
"The reality - and there is plenty of evidence globally to support this - is that most patients stop at one drug, often at low dose, without further increments," Patel said.
"This is called therapeutic inertia and probably driven by many factors - patients, doctors and the costs or the inconvenience of visiting doctors frequently to have drugs adjusted," she said. "The triple pill strategy essentially bypasses therapeutic inertia. We believe this will be a highly cost-effective approach."
The clinical trial had initially been planned and funded to be conducted in India. "We had to move to Sri Lanka after repeated failure to obtain approval (from drug regulators)," Patel said.
The rejections had come at a time new clinical trials had been stalled amid concerns that some earlier trials had breached ethics.