 Tuesday, 07 May 2024
Tuesday, 07 May 2024
 Tuesday, 07 May 2024
Tuesday, 07 May 2024
















Big B and Rajnikanth have joined hands for ‘Vettaiyan’ after 1991 hit ‘Hum’
Gear up for Baahubali’s animated spin-off
Get trendy with this Ayushmann Khurrana-approved lifestyle brand
Don’t miss Nussrat Jahan’s hilarious attempt at using chopsticks
Beat the heat with chef Kunal Kapur’s Rose Mojito recipe
Israel is not the only country where Indian workers face dire working situations. Exploitation of Indian workers under conditions akin to slavery in Singapore & Gulf states has been a long-standing issue
CAROL SCHAEFFER
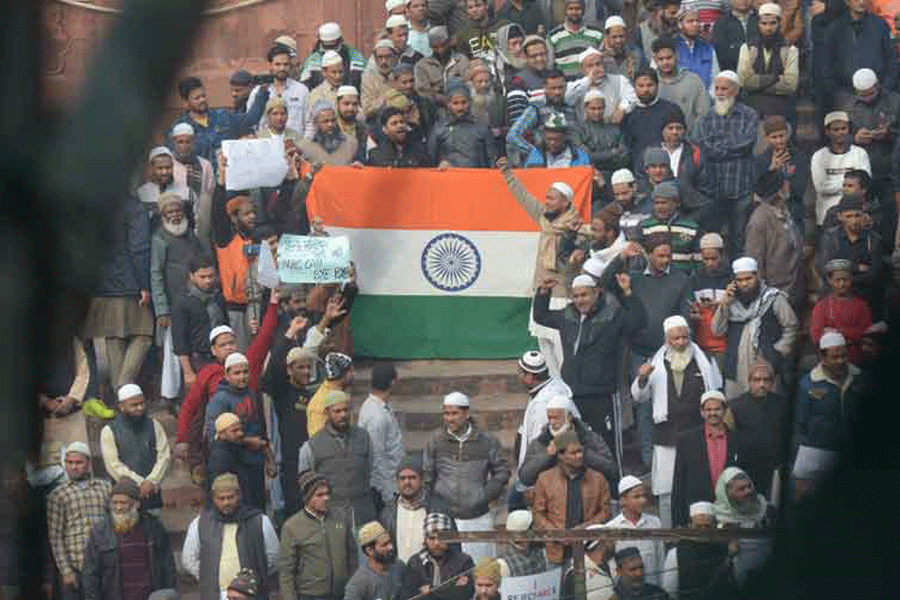
The inclusionary spirit of nation-building was distilled in the Constitution that directs the State to protect equality and non-discrimination that nurture the nation’s diversity
SUHIT K. SEN
Let us reflect on some hard realities of India. ‘Mere’ corruption is no longer an electoral issue: we do not shun politicians because they might have their hands in the till or in our pockets
SUKANTA CHAUDHURI
Epidemic Plan, envisioned as a collaborative endeavour among the Central govt, state governments, and relevant stakeholders, aims to provide a comprehensive blueprint for coordinated action
BASIL GUPTA
Intelligence-sharing has been at the heart of India’s deepened strategic ties with US & its allies. Such exchange of information relies on mutual trust between intelligence agencies
THE EDITORIAL BOARD
































Starring Monisha Koirala, Sonakshi Sinha, Aditi Rao Hydari and Richa Chadha, the eight-episode Heeramandi is streaming on Netflix






















